Inhibitor of Phosphatidylinositol Turnover Inostamycin A (sodium salt)
Date:July 03 2020Web Page No:520050
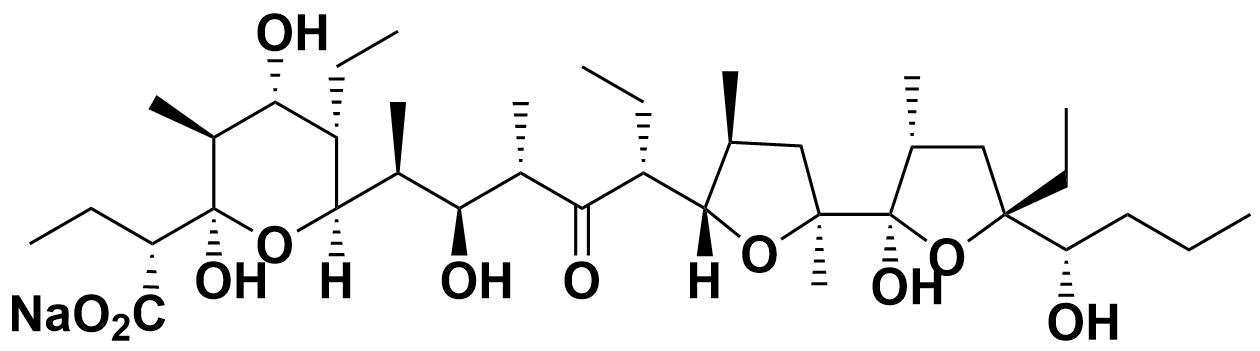
Inostamycin A is isolated from the culture fluid of Streptomyces sp. MH816-AF15 and inhibits cytidine diphosphodiacylglycerol (CDP-DG): inositol transferase 1).
Inostamycin A inhibited the incorporation of inositol into inositol lipids induced by EGF in an assay system using A431 cells, with 50 an IC50 value of approximately 0.5 μg/ml 2).
The IC50 of in vitro CDP-DG in A431 cell membranes was approximately 0.02 μg/ml 1).
It exhibited cytolytic activity against srk-NIH-3T3 cells (IC50: approximately 0.07 μg/ml) 3).
Inostamycin A at 3 μg/ml enhanced the cytotoxicity of colchicine against KB-C4 cells 4).
0.1 μg/ml caused accumulation of MS-1 cells into the G1 phase, while a higher concentration (0.3 μg/ml) induced morphological apoptosis 6).
It has also been reported that it enhances the cell-killing effect of taxol against small cell lung cancer cells 5).
* Please do not hesitate to contact Funakoshi if you need a SDF (Structure Data Format) file and/or bulk purchase (large quantity) of the compounds.
* This product is for research use only.
Specification
| CAS# | 1884611-95-3 |
|---|---|
| Molecular Formula | C38H67O11 Na |
| Molecular Weight | 722.933 |
| Source | Streptomyces sp. MH816-AF15 |
| Purity | > 90% (Quantitative NMR) |
| Solubility | Soluble in DMSO, DMF and CHCl3. Poorly soluble in H2O |
Mechanism
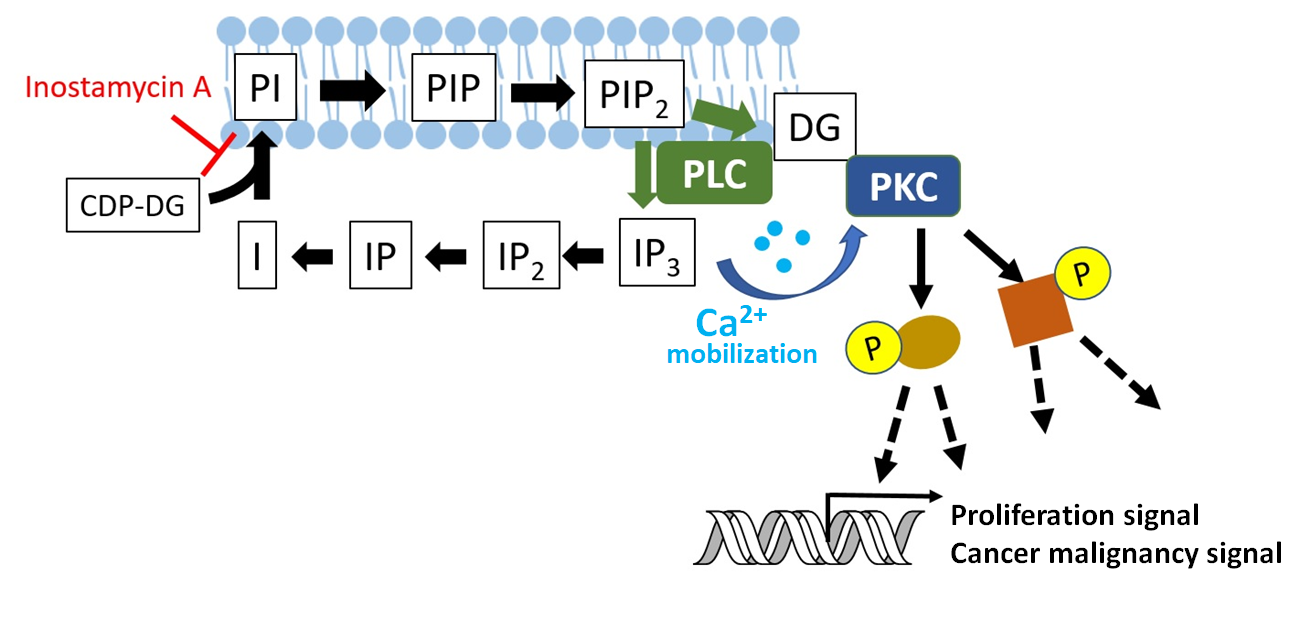
Phosphatidylinositol 2-phosphate (PIP2), generated from phosphatidylinositol (PI) present in the cell membrane, is degraded by phospholipase C (PLC) to inositol triphosphate (IP3) and diacylglycerol (DG), and IP3 is converted to inositol (I). The inositol (I) is bound to CDP-DG and PI is resynthesized. This metabolic turnover is called the inositol phospholipid turnover. IP3 generated by this turnover releases Ca2+ from the endoplasmic reticulum, and the Ca2+ and DG activate protein kinase C (PKC), which phosphorylates various intracellular proteins to activate proliferation signals and cancer malignancy signals.
Inostamycin A inhibits the synthesis of PI from I and CDP-DG in inositol-phospholipid-turnover and arrests cell-cycle in G1 phase, thereby inhibiting cell proliferation 2).
Application Note
- Inhibited in vitro CDP-DG: inositol transferase activity of the A431 cell membrane, the IC50 was about 0.02 μg/ml 1)
- Inhibited EGF-induced inositol incorporation into inositol lipids with an IC50 of about 0.5 μg/ml in the A431 cell assay system 2)
- Showed antimicrobial activities against Gram-positive bacteria and cytocidal activity against srk-NIH-3T3 cell with IC50 value of 0.07 μg/ml 3)
- Potenciated colchitine cytotoxicity toward KB-C4 cells at 3 μg/ml 4)
- Enhanced the cytotoxic effect of Taxol on small cell lung cancer cells 5)
- Caused Ms-1 cell to accumulate in the G1 phase at 0.1 μg/ml and induces morphological apoptosis at high concentration (0.3 μg/ml) 6)
- It irreversibly binds to the cell membrane of multidrug-resistant human cancer cell line KB, and irreversibly inhibits P-glycoprotein through specific interaction with phospholipid phosphatidylethanolamine 7).
- When the concentration of inostamycin in the culture medium was 62.5~125 ng/ml, cell proliferation of the human oral squamous cell carcinoma cell line used in the study was inhibited 8).
* Biological activity of the product was not verified. The values shown above were cited from the references.
Reference
- Imoto M., et al., J. Biochem., 112, 299-302(1992). [PMID:1328172]
- Imoto M., et al., J. Nat. Prod., 53(4), 825-829(1990). [DOI:10.1021/np50070a008]
- Odai H., et al., J. Antibiot., 47(8), 939-941(1994). [PMID:7928682]
- Kawada M., et al., J. Antibiot., 45(4), 556-562(1992). [PMID:1592687]
- Imoto M., et al., Jpn. J. Cancer Res., 89(3), 315-322(1998). [PMID:9600126]
- Simizu S., et al., Jpn. J. Cancer Res., 89(9), 970-976(1998). [PMID:9818034]
- Kawada, M. and Umezawa, K., Jpn. J. Cancer Res., 86(9), 873-878(1995). [PMID:7591966]
- Baba, Y., et al., Cell Biol. Int., 25(7), 613-620(2001). [PMID:11448100]

[Date : June 06 2026 00:08]
| Detail | Product Name | Product Code | Supplier | Size | Price | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Inostamycin A (Sodium Salt), Antitumor DatasheetThis may not be the latest data sheet. |
14652 | IMCInstitute of Microbial Chemistry | 1 mg | $500 | |||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||
[Date : June 06 2026 00:08]
Inostamycin A (Sodium Salt), Antitumor
DatasheetThis may not be the latest data sheet.
- Product Code: 14652
- Supplier: IMC
- Size: 1mg
- Price: $500
| Description |
M.W.: 722.933 (Sodium salt) Purity : >90% (qNMR) Molecular Formula : C38H68O11Na (Sodium salt) Solubility : Soluble in DMSO, DMF, and CHCl3, Poorly soluble in H2O. Inostamycin A is an inhibitor of phosphatidylinositol turnover, supplied as sodium salt. It was reported that this compound inhibited EGF-induced inositol incorporation into inositol lipids in the A431 cell assay system. It was also reported that inhibition of in vitro CDP-DG: inositol transferase activity of the A431 cell membrane, antimicrobial activities against gram-positive bacteria, cytocidal activity against srk-NIH-3T3, and potentiated colchicine cytotoxicity toward KB-C4 cells. In addition, it caused Ms-1 cell to accumulate in the G1 phase and induced morphological apoptosis at high concentration. |
||
|---|---|---|---|
| Storage | -20°C | CAS | 1884611-95-3 (sodium salt), 129905-10-8 (salt free form) |
| Link | |||
CONTACT
export@funakoshi.co.jp
- ※Prices on our website are for your reference only. Please inquire your distributor for your prices.
- ※Please note that Product Information or Price may change without notice.