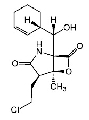
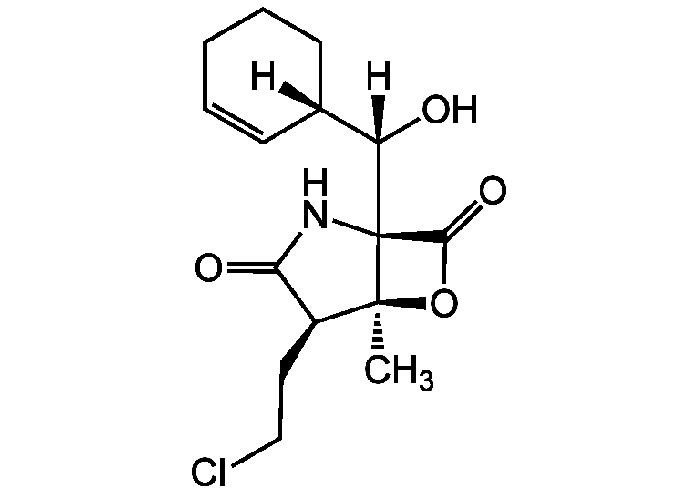
| 特長 | - Potent, irreversible inhibitor of all the 3 proteolytic activities of the mammalian 20S proteasome.
- β5 subunit: chymotrypsin-like (EC50 = 3.5nM)
- β2 subunit: trypsin-like (EC50 = 28nM)
- β1 subunit: caspase-like or peptidyl-glutamyl peptide-hydrolyzing (PGPH) (EC50 = 430nM)
- Potent anticancer compound.
- Triggers apoptosis, with distinct proteasome activity and mechanism of action compared to bortezomib (Velcade) (Prod. No. AG-CR1-3602).
- Most potent suppressor of NF-κB activation, compared with bortezomib, MG-132 (Prod. No. AG-CP3-0011), N-acetyl-leucyl-leucyl-norleucinal (ALLN) and lactacystin (Prod. No. AG-CN2-0104).
- Inhibitor of TNF-α, IL-1, IL-6, ICAM-1 and VEGF synthesis.
- Displays a longer inhibition duration than bortezomib.
- Potent antileukemic activity against bortezomib-resistant leukemia cells.
|
|---|
| 文献 | - Salinosporamide A: a highly cytotoxic proteasome inhibitor from a novel microbial source, a marine bacterium of the new genus salinospora: R.H. Feling, et al.; Angew. Chem. Int. Ed. Engl. 42, 355 (2003)
- Structure-activity relationship studies of salinosporamide A (NPI-0052), a novel marine derived proteasome inhibitor: V.R. Macherla, et al.; J. Med. Chem. 48, 3684 (2005)
- A novel orally active proteasome inhibitor induces apoptosis in multiple myeloma cells with mechanisms distinct from Bortezomib: D. Chauhan, et al.; Cancer Cell 8, 407 (2005)
- Crystal structures of Salinosporamide A (NPI-0052) and B (NPI-0047) in complex with the 20S proteasome reveal important consequences of beta-lactone ring opening and a mechanism for irreversible binding: M. Groll, et al.; JACS 128, 5136 (2006)
- NPI-0052 enhances tumoricidal response to conventional cancer therapy in a colon cancer model: J.C. Cusack, et al.; Clin. Cancer Res. 12, 6758 (2006)
- Comparison of biochemical and biological effects of ML858 (salinosporamide A) and bortezomib: M.J. Williamson, et al.; Mol. Cancer Ther. 5, 3052 (2006)
- Salinosporamide A (NPI-0052) potentiates apoptosis, suppresses osteoclastogenesis, and inhibits invasion through down-modulation of NF-κB-regulated gene products: K.S. Ahn, et al.; Blood 110, 2286 (2007)
- A mechanistic and kinetic study of the beta-lactone hydrolysis of Salinosporamide A (NPI-0052), a novel proteasome inhibitor: N. Denora, et al.; J. Pharm. Sci. 96, 2037 (2007)
- Combination of proteasome inhibitors bortezomib and NPI-0052 trigger in vivo synergistic cytotoxicity in multiple myeloma: D. Chauhan, et al.; Blood 111, 1654 (2008)
- Discovery and development of the anticancer agent salinosporamide A (NPI-0052): W. Fenical, et al.; Bioorg. Med. Chem. 17, 2175 (2009)
- Generating a generation of proteasome inhibitors: from microbial fermentation to total synthesis of salinosporamide a (marizomib) and other salinosporamides: B.C. Potts & K.S. Lam; Mar. Drugs 8, 835 (2010) (Review)
- Salinosporamide natural products: Potent 20S proteasome inhibitors as promising cancer chemotherapeutics: T.A. Gulder & B.S. Moore; Angew. Chem. Int. Ed. Engl. 49, 9346 (2010) (Review)
- Marizomib, a proteasome inhibitor for all seasons: preclinical profile and a framework for clinical trials: B.C. Potts, et al.; Curr. Cancer Drug Targets 11, 254 (2011)
- Proteasome regulator marizomib (NPI-0052) exhibits prolonged inhibition, attenuated efflux, and greater cytotoxicity than its reversible analogs: A. Obaidat, et al.; J. Pharmacol. Exp. Ther. 337, 479 (2011)
- Molecular mechanisms of acquired proteasome inhibitor resistance: A.J. Kale & B.S. Moore; J. Med. Chem. 55, 10317 (2012)
- Antileukemic activity and mechanism of drug resistance to the marine Salinispora tropica proteasome inhibitor salinosporamide A (Marizomib): D. Niewerth, et al.; Mol. Pharmacol. 86, 12 (2014)
|
|---|