HOME> Products> Antibody> Brain / Nerves> Anti-SOD1 (ALS-related mutants) Cocktail, Human, Rat-Mono (MS785/MS27)
Antibody cocktail for global detection of ALS-related SOD1 mutants Anti-SOD1 (ALS-related mutants) Cocktail, Human, Rat-Mono (MS785/MS27)
Date:October 15 2018Web Page No:81146

Funakoshi Co.,Ltd.
Our antibody cocktail (#FDV-0021A) contains two clones against SOD1 mutants. SOD1 mutants are thought to be one of the causes of Amyotrophic Lateral Sclerosis (ALS), and this cocktail specifically recognizes the three-dimensional structure commonly found in SOD1 mutants. The cocktail can detect more than 100 SOD1 mutants by immunostaining or western blotting after immunoprecipitation.
This antibody cocktail is also useful for SOD1 studies in zinc-deficient ER stress. ☞ Please click here for more information.
- Amyotrophic Lateral Sclerosis (ALS) and SOD1
- Features
- Application data
- Original Paper
- Product Information
Amyotrophic Lateral Sclerosis (ALS) and SOD1
Amyotrophic Lateral Sclerosis (ALS) is a neurodegenerative disease characterized by muscle weakness and atrophy of skeletal muscles throughout a body due to motor neuron cell death. About 90% of ALS cases are nonheritable, however about 10% are genetic, and about 20 genes have been identified as responsible genes. Among them, mutation of oxidoreductase SOD1 (Cu/Zn superoxide dismutase) is the most common cause of ALS, and transgenic mice expressing SOD1 mutants have been used as animal models of ALS. Pathophysiological mechanism of ALS caused by SOD1 mutations are still unclear, then it is expected to elucidate.
SOD1 is a metalloenzyme with copper and zinc ions in its active center, and degrades reactive oxygen species in cells. SOD1 is a relatively small protein of 154 amino acids (human) with 24 kDa, but more than 100 mutants have been reported (Fig. 1), and most of mutants have no effect on the enzyme activity that degrades reactive oxygen species. Therefore, it is believed that there are factors other than enzyme activity that contribute to the pathogenesis of ALS, and toxic gain of function associated with mutations is the most likely explanation.
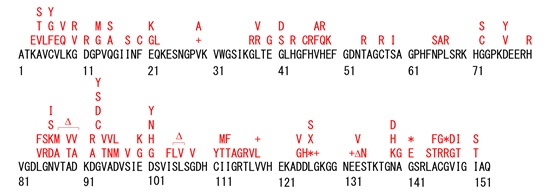
Fig. 1 Examples of reported SOD1 mutation sites
Although wild-type SOD1 originally forms dimers, Professor Hidenori Ichijo and co-workers have clarified the following points about SOD1 mutants:
- More than 100 different SOD1 variants share a common conformation that differs from wild type (Fig. 2).
- SOD1 mutants acquire cytotoxicity by binding Derlin-1, a component protein of ER-associated degradation (ERAD) machinery protein in endoplasmic reticulum.
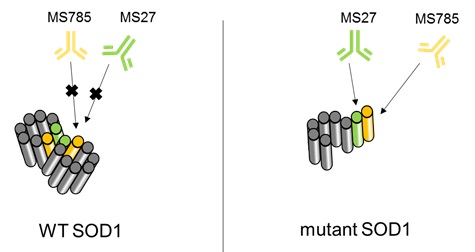
Fig. 2 Structure difference and antibody recognition site between wild-type and SOD1 mutant
Furthermore, they succeeded in developing rat monoclonal antibodies clone MS785 and MS27, which specifically bind to the three-dimensional structure found in SOD1 mutants. These antibodies are expected to be an excellent tool for investigating pathophysiological roles of SOD1 mutants in ALS patients.
Features
- Antibody cocktail of SOD1 rat monoclonal antibody, clone MS785 and MS27
※ Clone MS785 and MS27 can be purchased separately. - Clones MS785 and MS27 recognize monomeric conformation commonly found in SOD1 mutants. More than 100 SOD1 mutants have been detected after immunoprecipitation. ☞ Please click here for validated SOD1 mutants.
- It can detected SOD1 mutants specifically by immunocytochemistry.
- It can detect conformational changes in wild-type SOD1 under zinc-deficient conditions, which may be one cause of endoplasmic reticulum stress. It is also useful for endoplasmic reticulum stress research and basic research on SOD1. ☞ Please click here for more information.
- Application : Immunoprecipitation & Immunostaining: detects SOD1 mutants, ELISA: detects SOD1 mutants (for details, please see ☞ Original Paper 2), Western blot: detects both SOD1 mutants and wild type
※ Note : In the denatured condition, not only SOD1 mutants but also wild-type are detected. Please be careful to avoid protein denaturation and do not use denaturing agents such as SDS.
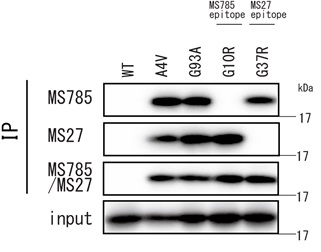
Difference between clone MS785 and MS27
| Clone | MS785 | MS27 |
|---|---|---|
| Type | Rat monoclonal antibody | |
| Subtype | IgG2b/κ | IgG2a/κ |
| Antigen | Human SOD1 6-16 amino acids | Human SOD1 30-40 amino acids |
| Reactivity | Human | |
| Fromat | Protein G purified (0.5 mg/ml), Buffer: 50% glycerol / PBS | |
- Both clones can detect a wide range of SOD1 mutants, however they cannot detect variants with mutations in each epitope.
- Because of the complementarity between MS785 and MS27, the cocktail antibody can complement detection of epitope variants.
Application data
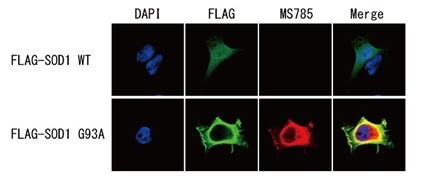
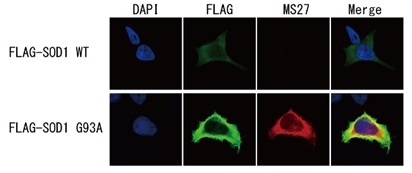
Detection of SOD1 mutants by immunocytochemistry
Flag-tagged SOD1 wild type (WT) or G93A mutant were expressed in HEK293 cells, then immunostained with 1μg/mL MS785, MS27 or MS785/MS27 cocktail (Red) and FLAG antibody (Green).
Both wild-type and G93A mutant were seen with FLAG antibody, but neither MS785, MS27, nor MS785/27 cocktail reacted with the wild-type but only with mutant.
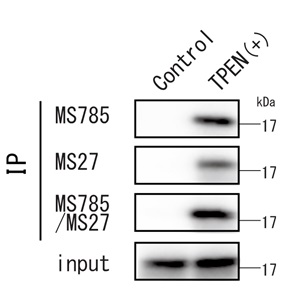
Zinc deficiency and SOD1 conformational change
Reference
- Fujisawa, T., et al., "A novel monoclonal antibody reveals a conformational alteration shared by amyotrophic lateral sclerosis-linked SOD1 mutants.", Ann. Neurol.,72(5), 739~749(2012). [PMID:23280792]
- Homma, K., et al.,"SOD1 as a Molecular Switch for Initiating the Homeostatic ER Stress Response under Zinc Deficiency.", Molecular Cell, 52(1), 75~86 (2013). [PMID:24076220]
- Fujisawa, T., et al.," A systematic immunoprecipitation approach reinforces the concept of common conformational alterations in amyotrophic lateral sclerosis-linked SOD1 mutants.", Neurobiol. Dis., 82, 478~486(2015). [PMID:26297318]
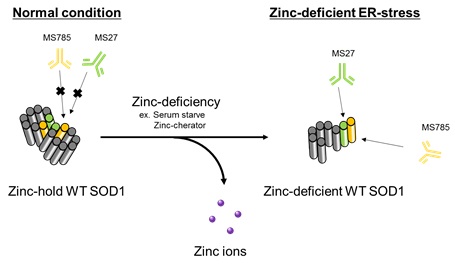
Detection of endogenous SOD1 wild type under ER-stress
This antibody can detect dynamic conformational changes in wild-type SOD1 during zinc deficiency. It is useful not only for ALS research but also for basic SOD1 research.
- Endogenous wild-type SOD1 has been detected in serum deficient conditions or when treated with the zinc chelator TPEN.
- Detectable with MS785, MS27 or the cocktail.
Flag-tagged SOD1 wild type (WT) or G93A mutant were expressed in HEK293 cells, then immunostained with 1μg/mL MS785, MS27 or MS785/MS27 cocktail (Red) and FLAG antibody (Green). Both wild-type and G93A mutant were seen with FLAG antibody, but neither MS785, MS27, nor MS785/27 cocktail reacted with the wild-type but only with mutant.
Zinc deficiency and SOD1 conformational change
Reference
- Fujisawa, T., et al., "A novel monoclonal antibody reveals a conformational alteration shared by amyotrophic lateral sclerosis-linked SOD1 mutants.", Ann. Neurol.,72(5), 739~749(2012). [PMID:23280792]
- Homma, K., et al.,"SOD1 as a Molecular Switch for Initiating the Homeostatic ER Stress Response under Zinc Deficiency.", Molecular Cell, 52(1), 75~86 (2013). [PMID:24076220]
- Fujisawa, T., et al.," A systematic immunoprecipitation approach reinforces the concept of common conformational alterations in amyotrophic lateral sclerosis-linked SOD1 mutants.", Neurobiol. Dis., 82, 478~486(2015). [PMID:26297318]
Detection of endogenous SOD1 wild type under ER-stress
This antibody can detect dynamic conformational changes in wild-type SOD1 during zinc deficiency. It is useful not only for ALS research but also for basic SOD1 research.
- Endogenous wild-type SOD1 has been detected in serum deficient conditions or when treated with the zinc chelator TPEN.
- Detectable with MS785, MS27 or the cocktail.
Original Paper
- Fujisawa, T., et al., "A novel monoclonal antibody reveals a conformational alteration shared by amyotrophic lateral sclerosis-linked SOD1 mutants.", Ann. Neurol.,72(5), 739~749(2012). [PMID:23280792]
- Fujisawa, T., et al.," A systematic immunoprecipitation approach reinforces the concept of common conformational alterations in amyotrophic lateral sclerosis-linked SOD1 mutants.", Neurobiol. Dis., 82, 478~486(2015). [PMID:26297318]
Product Information
[Date : May 31 2026 00:07]
| Detail | Product Name | Product Code | Supplier | Size | Price | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Anti-SOD1(ALS-related mutants) Cocktail, Human, Rat-Mono (MS785/MS27) DatasheetThis may not be the latest data sheet. |
FDV-0021A | FNAFunakoshi Co.,Ltd. | 100 µg | $400 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Anti-SOD1(ALS-related mutants), Human, Rat-Mono (MS785) DatasheetThis may not be the latest data sheet. |
FDV-0021B | FNAFunakoshi Co.,Ltd. | 100 µg | $400 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Anti-SOD1(ALS-related mutants), Human, Rat-Mono (MS27) DatasheetThis may not be the latest data sheet. |
FDV-0021C | FNAFunakoshi Co.,Ltd. | 100 µg | $400 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
[Date : May 31 2026 00:07]
Anti-SOD1(ALS-related mutants) Cocktail, Human, Rat-Mono (MS785/MS27)
DatasheetThis may not be the latest data sheet.
- Product Code: FDV-0021A
- Supplier: FNA
- Size: 100µg
- Price: $400
| Description |
New powerful tools to research pathophysiological role of SOD1 mutants in ALS patients |
||
|---|---|---|---|
| Storage | -20°C | ||
| Antigen Species | Human | Host Species | Rat |
| Class | IgG | Label | |
| Cross Reactivity | Human | Application | IP,IC,ELISA,Western Blot |
| Clonality | Format | ||
| Purification | Affinity Purified | Absorption | |
| Link |
|
||
Anti-SOD1(ALS-related mutants), Human, Rat-Mono (MS785)
DatasheetThis may not be the latest data sheet.
- Product Code: FDV-0021B
- Supplier: FNA
- Size: 100µg
- Price: $400
| Description |
Powerful tools to research pathophysiological role of SOD1 mutants in ALS patients |
||
|---|---|---|---|
| Storage | -20°C | ||
| Antigen Species | Human | Host Species | Rat |
| Class | IgG | Label | |
| Cross Reactivity | Human | Application | IP,IC,ELISA,Western Blot |
| Clonality | Format | ||
| Purification | Affinity Purified | Absorption | |
| Link |
|
||
Anti-SOD1(ALS-related mutants), Human, Rat-Mono (MS27)
DatasheetThis may not be the latest data sheet.
- Product Code: FDV-0021C
- Supplier: FNA
- Size: 100µg
- Price: $400
| Description |
Powerful tools to research pathophysiological role of SOD1 mutants in ALS patients. |
||
|---|---|---|---|
| Storage | -20°C | ||
| Antigen Species | Human | Host Species | Rat |
| Class | IgG | Label | |
| Cross Reactivity | Human | Application | IP,IC,ELISA,Western Blot |
| Clonality | Format | ||
| Purification | Affinity Purified | Absorption | |
| Link |
|
||
CONTACT
export@funakoshi.co.jp
- ※Prices on our website are for your reference only. Please inquire your distributor for your prices.
- ※Please note that Product Information or Price may change without notice.